Table of Contents
Drug Class
H2 receptor antagonist (H2 blocker)
Antiulcer drug
Reduces gastric acid secretion
H2 receptors are present in:
- Stomach parietal cells
Brand Names
India:
- Rantac
- Aciloc
- Zinetac
USA
- Zantac (discontinued)
UK / Europe:
- Zantac
Approved By
- FDA (United States)
- CDSCO (India)
- EMA (Europe)
⚠️ Note: Ranitidine has been withdrawn or restricted in many countries due to NDMA impurity concerns.
Available Strengths & Dosage Forms
- Tablet: 150 mg, 300 mg
- Syrup: 75 mg / 5 ml
- Injection: 25 mg / ml
Purpose / Indications
It is used to reduce stomach acid and treat acid-related disorders.
Symptoms Ranitidine May Cure
- Acid reflux
- Heartburn
- Peptic ulcer
- GERD (Gastroesophageal reflux disease)
- Gastritis
- Zollinger-Ellison syndrome
Dosage
Adults
- GERD / Ulcer:
150 mg twice daily
Or
300 mg once daily
Children
- Dose depends on body weight and physician prescription
⚠️ Dose should be adjusted as directed by a physician.
Mechanism of Action
Ranitidine blocks H2 receptors in gastric parietal cells
This leads to:
Reduced acid secretion
Reduced gastric volume
Reduced acidity
Result: Relief from ulcer and reflux symptoms
Detailed Explanation
Let’s first understand the role of histamines in stomach acid production, then we will be able to understand role of antihistamines in prevention of stomach acid production.
Role of histamine in stomach acid production
H2 receptors are present on the surface of gastric parietal cells in the stomach lining. These receptors regulate acid secretion. When histamine binds to H2 receptors, it activates parietal cells and stimulates the secretion of hydrochloric acid.
Role of Ranitidine in prevention of excessive stomach acid production
Ranitidine blocks these H2 receptors, preventing histamine from activating acid production. This reduces gastric acid secretion and helps treat conditions such as GERD, peptic ulcer, and acid reflux.
In one Line: H2 receptors are acid control switches on parietal cells, and Ranitidine blocks these switches to reduce acid production.

Additional pharmacological points
Bioavailability
Bioavailability refers to the percentage of a drug that reaches systemic circulation in an active form after administration.
Bioavailability of ranitidine is Approximately 50%
Half life
Half-life is the time required for the plasma concentration of a drug to reduce to half of its original value.
Half-life of ranitidine is 2–3 hours
Summary statement:
Ranitidine is an H₂ receptor blocker that reduces stomach acid production. In the stomach, histamine normally binds to H₂ receptors on parietal cells and stimulates acid secretion. Ranitidine blocks these H₂ receptors, preventing histamine from activating acid release. As a result, gastric acid secretion decreases, which helps treat conditions such as GERD, peptic ulcers, and acid reflux.
Side Effects
Common Side Effects
- Headache
- Dizziness
- Constipation
- Diarrhea
Less Common / Rare
- Confusion (elderly)
- Bradycardia
- Hypersensitivity
Contraindications
- Known hypersensitivity.
- Severe renal impairment (dose adjustment may be required)
Use in Special Populations
Pregnancy
- Generally considered safe (Category B– FDA older classification)
- Use only if clearly needed and prescribed by a physician
A drug is placed in Category B when animal reproductive studies have not shown a risk to the fetus, but there are no adequate and well-controlled studies in pregnant women. That means:
- Animal studies: Did not show evidence that the medicine causes fetal harm.
- Human data: Not enough good studies yet to confirm whether it’s absolutely safe or risky.
So Category B suggests the medicine may be safer than other categories, but this is not a guarantee—doctors still use it only if benefits outweigh potential risks.
Lactation
- Passes into breast milk
- Use cautiously
Elderly
- Usually safe
- Dose adjustment may be required in renal impairment
Drug Interactions
May interact with:
- Warfarin
- Ketoconazole
- Other acid-reducing drugs
Important Safety Note
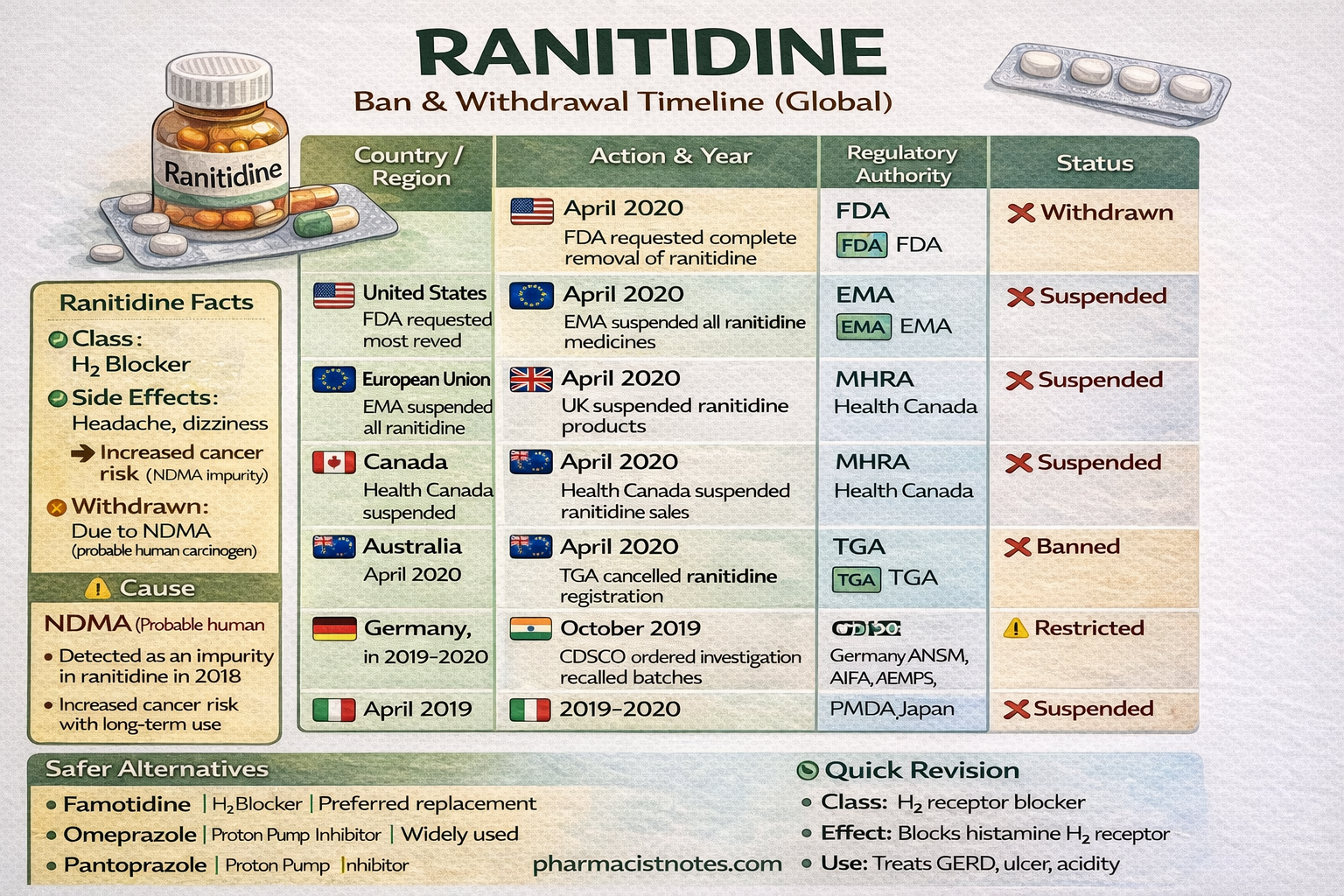
Ranitidine has been withdrawn in many countries due to contamination with NDMA, a probable carcinogen.
| Country / Region | Regulatory Authority | Year | Current Status |
|---|---|---|---|
| United States | FDA (Food and Drug Administration) | April 2020 | ❌ Withdrawn / banned |
| European Union | EMA (European Medicines Agency) | April 2020 | ❌ Suspended |
| United Kingdom | MHRA (Medicines and Healthcare products Regulatory Agency) | April 2020 | ❌ Suspended |
| Canada | Health Canada | April 2020 | ❌ Suspended |
| Australia | TGA (Therapeutic Goods Administration) | April 2020 | ❌ Banned |
| Germany | BfArM | 2020 | ❌ Suspended |
| France | ANSM | 2020 | ❌ Suspended |
| Italy | AIFA | 2020 | ❌ Suspended |
| Spain | AEMPS | 2020 | ❌ Suspended |
| India | CDSCO (Central Drugs Standard Control Organization) | 2019 | ⚠️ Restricted / limited use |
| Japan | PMDA | 2020 | ❌ Suspended |
| Switzerland | Swissmedic | 2020 | ❌ Suspended |
| Cause | Details |
|---|---|
| Contaminant | NDMA (N-Nitrosodimethylamine) |
| Risk | Probable human carcinogen |
| Problem discovered | 2018 |
| Major withdrawal year | 2020 |
Other safer alternatives include:
Famotidine
Omeprazole
Storage Instructions
- Store at room temperature
- Keep away from moisture and direct sunlight
- Keep out of reach of children
Disclaimer
This information is for educational purposes only and should not be used as a substitute for professional medical advice. Always consult a doctor or pharmacist before using any medication.
Key points / Revision
| Point | Details |
|---|---|
| Drug Class | H2 blocker |
| Mechanism | Blocks H2 receptors |
| Use | GERD, ulcer |
| Acid secretion | Reduced |
| Duration | 8–12 hours |
| Half-life | 2–3 hours |
| Major concern | NDMA impurity |
| Alternative | Famotidine |


One thought on “RANITIDINE: Uses, Dosage, Side Effects, Contraindications & Safety”